Updates!
Prospective students should email me (matthew.kolmann@louisville.edu) if they are interested in graduate student positions studying morphology, ecology & evolution.
Follow me on Twitter @KolmannMA and through Google Scholar!
Finally, we understand the evolutionary relationships among piranhas & their herbivorous cousins, pacus
our new paper in Systematic Biology

Read the whole thing here.
What is a pacu, anyway? Pacus are the herbivorous cousins of piranhas. They are some of the largest fishes in the Amazon and also serve as several of the primary seed dispersers for trees that live in the flooded forest or varzea.
What's so contentious about pacu and piranha relationships anyway? Well, a couple things:
(1) there are 16 genera and just under 100 species in Serrasalmidae (the family that includes pacus and piranhas). There have been some great studies on the relationships of these genera in the past, but no studies have included all extant genera, nor did they resolve relationships among major groups.
(2) When did piranhas and pacus evolve? This has been extra contentious, since we rely on very scant fossil data to make inferences about the ages of these groups. Some estimates are based on isolated fossil teeth that look suspiciously like other characiforms...
(3) What evolved first, herbivory or carnivory? Or rather, what did the ancestor of modern piranhas look and act like, and moreover, what did the common ancestor of all serrasalmids look like? We need a modern, comprehensive phylogeny (a hereditary hypothesis for the relationships among species and genera) to ask these questions.
What did we find?
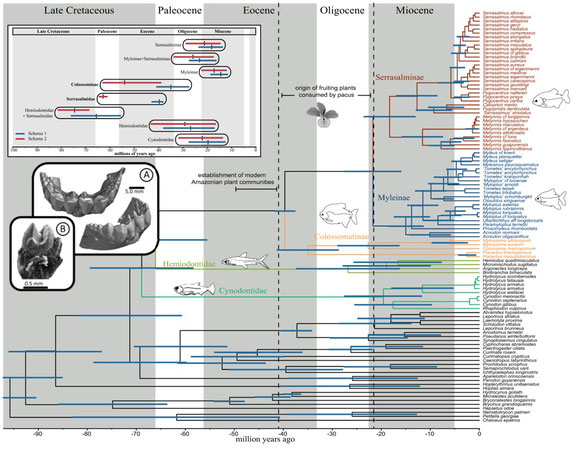
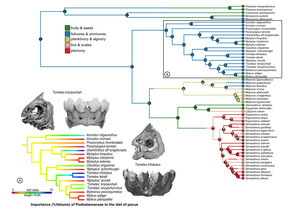
(1) Our study is the first to confidently resolve the relationship of all extant genera. The largest pacu species are now in a subfamily called Colossomatinae, while the vast majority of pacus are in the subfamily Myleinae. Piranhas are sister to silver dollars (Metynnis) in Serrasalminae. We found that many valid genera are probably not so valid... the taxonomy of Myleinae in particular is in need of major revision. We resuscitated the names Paramyloplus and Prosomyleus for species that are distinctive enough from other myleines, but almost all other genera in Myleinae warrant revision.
(2) The ancestor of all pacus and piranhas evolved around 42-38 million years ago (mya), solidly within the Eocene. Interestingly, this timeline fits with the diversification of many of the plant families that large pacus like Colossoma or tambaqui, feed on today. So bigger pacus have been around for a long, long time - while the more diverse myleines (e.g., Myloplus, Tometes, etc.) are more recent. Piranhas evolved in the Miocene, less than 20-15 mya - but most of modern piranha species diversity arose in the last 5 million years.
(3) The most ancient serrasalmids were pacu-like and ate fruits or leaves of riparian plants. The ancestor of all modern piranhas was probably herbivorous too, to some extent at least, and probably fed on fruits, seeds, insects, and the fins or scales of other fishes. Modern piranhas like Pygopristis still do this, and some smaller pacus, like Acnodon do as well. In fact, a lot of the shorter snouted Serrasalmus and Pristobrycon still feed on fruits and seeds at certain times during the year. Fruit-feeding (frugivory) seems to be flexible, surplus sort of resource for serrasalmids through their entire history. There's also unappreciated diversity within 'herbivores,' with species specializing on everything from encrusting bryophytes in rapids, to crushing seeds, to feeding on algae in impounded lagoons and river ox-bows.
How to build strong, yet flexible armor: inspiration from weird fishes with armored eyeballs
our new paper in the Journal of the Royal Society: Interface

How can we learn to make better, harder, faster, stronger... stuff? Read the story here.
One answer is looking to biological examples for inspiration. Afterall, adaptation is an iterative process that refines the workings of organisms over millions of years.
We studied the armor of regrettably-named 'poacher' fishes, the Agonidae. Poachers are not as bad as their name suggests, they are cute armored fishes that make a relatively low-key living along the ocean floor in the northern Pacific and even into the Arctic and North Atlantic. They are notable because every species is armored (and there are just under 50 species), with some incredible diversity of armor to boot. Some species are entirely covered, with openings in the armor only their mouths, eyeballs, and anus... and yet some taxa even have small armored scales on their eyeballs too.
We filmed poacher species swimming in little track-like arenas, measured how well their bodies bent, and used CT scanning and electron microscopy to characterize what their armor looked like and how it fit together. Turns out that poachers are a GREAT example of how to build strong, yet flexible structures. The overlapping armor plates lend stiffness to the body, and while this heavier, denser armor slows down adult poachers (juveniles are still pretty quick), the greater stiffness of the armor makes for faster accelerations in adults.
Stay tuned for more poacher work - we're hoping to get insight into how to build armored articulated gantries or arms for deepwater ROVs or submarines, or even how to learn to make flexible, tiled surfaces.
Fight it or Flaunt it: electric fish battle with long faces
our new paper in the Zoological Journal of the Linnean Society

What is the elongate snout of some ghost knifefishes (Apteronotidae) for? Why do male and female fishes look different?
Does Fish Fight Club exist? Some answers below...
Q. Static allometry? Sexually dimorphic? Phenotype? What does it all mean?!
A: Scientists have long been interested in why there’s such a huge diversity of life on Earth; questions spanning a continuum from what distinguishes different species to why members of the opposite sex look so different from one another in one species, but not another. As such, we use very specific terms to describe the particular kind of shapes we observe, as well as the patterns we see in how certain shapes are represented across entire groups of animals. So, when we say ‘phenotype,’ we mean the overall shape and character of an animal, as a contrast to its genotype, the genetic blueprint underlying the phenotype. Studies of ‘sexual dimorphism’ describe differences in shape and behavior between the sexes, in this case how male electric knifefishes have elongated snouts. Static allometry, describes a pattern of how shape changes as an animal grows and develops from juvenile to adulthood, specifically how one aspect of an animal, let’s say the snout, can grow out of proportion with the rest of the body.
Q. Wait, why would one part of the body grow out of proportion to the rest?
A: That’s the million(>s)-year old question! Scientists generally propose that when one trait (or a couple related traits) grow out of proportion to the rest of the body, it suggests something bizarre is going on. Perhaps if traits are getting larger, faster, (cue Daft Punk) it might signify the importance of that trait to something the animal does – in this case, long snouts seem to be useful for fighting other males in electric fishes. Specifically, it’s long been proposed that these size-exaggerated traits, in the context of sexual dimorphism, show where sexual selection is acting on the evolution of a trait. Selection is the unseen (but measurable) force driving organisms to change their phenotype to adapt to changing conditions. Sexual selection is particularly weird, because it is selection acting differently between males and females in the same species.

Q: So electric fish snouts are like a baboon’s big blue butt?
A: Not to be too cheeky, but that’s a mandrill, not a baboon – baboons have big red butts. But anyway, yes – big colorful butts are a means for some apes to communicate their mating awesome-ness to rivals and potential mates alike. These are sexually-selected traits that drive males and females to look different from one another, and often this sort of selection will keep driving differences to become more and more exaggerated as thousands or more like millions of years go by. But while these traits may become bigger and bigger, such drastic changes in shape can alter their original function. Let’s take knifefishes for example: these are fishes that live in dark, murky freshwaters of South America like the Amazon River. They produce electric fields to sense their way around, find food, and communicate with other knifefishes. In one family, the Apteronotidae or ghost knifefishes, we found that males evolved longer snouts than females at least six times across the family. Males of some species have been seen fighting, biting and twisting with their elongated snouts – so we were interested in whether a knifefish species with one of the longest snouts (Compsaraia) was doing the same thing as other species.
Q. Why don’t electric fishes fight with, well… electricity?
A: Not a bad question – but as best we can tell, no. Electric eels are a kind of knifefish (don’t get us started on why it’s called an ‘eel’) and the only knifefish species that can injure other animals with its particularly potent electric discharges. However, many knifefishes broadcast different kinds of signals that communicate their rivalries, receptiveness to mating, or a simple ‘Is anyone out there? Does anyone really care?’ Sometimes you’ll find several species in one spot, and each of these species has a distinct signal to tell everyone apart, not to mention that males during the breeding season also have specific signals they use to let females know that they’re ready, willing (knifefish tinder perhaps?), and way better than the rest of those other male knifefish (totally, you can trust them). You can imagine that, for example, since brighter light bulbs tend to require more electricity, so do some electric signals – and so some males spend a lot of energy displaying to females – another example of sexual selection acting on a trait in knifefishes. But we digress…
Q. So seriously, why the long face?
A: We found that in two species of knifefishes, males grew longer snouts than females of the same species, and did this considerably faster as they grew. But something strange and yet not entirely unexpected came out of the data too... In Compsaraia samueli, a particularly long-snouted fish, we found that as males’ snouts elongate this lessened their ability to shut their mouths forcefully. CT scans also showed their snouts to be pretty weak, made of only thin bone. These fishes were elongating their faces… but it didn’t appear to be an effective weapon. Videos of two males sparring show that like skinny sumo wrestlers, males lock jaws during a sort of tug-of-war, and try to push each other out of an arena. Compare this to another long-snouted knifefish, Apteronotus rostratus, which have been reported as biting, pushing, twisting, and death-rolling with opponents. Apteronotus faces are also elongated, but males and females have similar jaw leverage, suggesting that both sexes maintain similar abilities as they develop. We think that while Apteronotus are using their jaws as a weapon to spar with rivals, Compsaraia snouts seem to function as more of a ritualized display. More of an ornament than a weapon - perhaps like peacock feathers used to signal to mates and potential rivals. So to answer your question of “why the long face?” The answer is: for fighting or flaunting, it depends on the species.
How stingrays eat clams made of harder stuff
our paper in Journal of Zoology on ray feeding biomechanics.

Q. Stingrays eat hard stuff with soft skeletons? What’s that about?
A: Elasmobranchs (sharks and rays) have skeletons made not of bone, like humans and other animals, but of cartilage. We have cartilage in our skeletons too, but usually constrained to places where we want the skeleton to be flexible (tip of your nose, your ears) or for use as a shock-absorbers (caps of cartilage in your joints). Cartilage skeletons give sharks and rays very flexible, very tough skeletons - but perhaps not stiff enough for crushing prey with shells, like clams and crabs. Turns out that elasmobranch jaws are stiffened by little mineralized tiles called tesserae that form a rind around the jaw. Some rays even have little mineralized struts called trabeculae inside their jaws that help distribute stresses that occur while these rays are chewing on shelled prey. So, as it turns out, sharks and rays have generally flexible skeletons, but mineralize certain parts of their skeleton where they need to, particularly the jaws. It’s a good thing too - durophagy (eating ‘hard’ stuff, prey with exoskeletons) has evolved many times throughout sharks, rays, and ratfishes.
Q. What is ontogeny?
A: Ontogeny is the process that occurs when animals change shape (development) and size (growth) as they age. Why not just say growth and development then? Changes in size can be distinctive from changes in shape. In biology we have some specialized terms so when scientists communicate with each other, they know exactly what the other means. Animals change their ecologies across ontogeny, sometimes because they can’t quite perform as well as adults, or because it’s easier to eat something else (and maybe avoid competing with their older relatives).
Q. How do you calculate bite forces?
A: We took stingrays captured through fisheries and dissected their jaw muscles and jaws. We measured where these muscles attached to the skeleton, how large the muscles are (and how muscle fibers are oriented), where the jaw joints were. From this we can estimate how much force the muscles can produce when they contract, where that force is being applied to the skeleton, and then by imagining the jaw as a lever (imagine a door frame, and where the best place is for your hand to close the door) we can get an idea of how much of the muscle force is transmitted through the jaws to the teeth. In some previous papers we’ve validated this bite force model by measuring bite forces in live stingrays - and it turns out - both methods give pretty similar results! Getting animals to behave in captivity is tough, so this way we can work with critters that have already been collected, even estimate bite forces in museum specimens that were collected hundreds of years ago!

Q. So animals don’t eat the same thing, no matter where they live?
A: It all depends on what’s available. When you’re hungry, you won’t forgo a hamburger when you really want a hot dog. There are fewer species of big, burrowing clams in the Gulf of Mexico than in Chesapeake Bay, so Florida cownose rays make do with a lot of other kinds of prey like little crustaceans, even sand dollars, in addition to surf clams and razor clams. But in Chesapeake Bay, cownose rays may even specialize on feeding on soft clams like Mya as well as larger species of razor clams. Turns out though, that despite really high bite forces in Chesapeake Bay cownose rays, even big oysters, clams, and cockles are still too strong for rays to bite into.
Q. Are there other stingrays that eat ‘hard’ stuff?
A: There are! There are at least five separate instances of durophagy that evolve in batoids (rays, skates, and guitarfishes). Some freshwater rays from the Amazon can eat shelled prey like snails and crabs (Potamotrygon sp.), as well as weird guitarfishes like Rhina, and big whiprays like Pastinachus in Australia and the Indo-Pacific. The entire Myliobatidae family - eagle rays, bat rays, and cownose rays - all these species eat shelled mollusks or crustaceans as their primary prey. What’s sort of weird however is that the nearest cousins to the cownose rays are the mobulids (mantas and devil rays) which are mostly planktivores like whales, whale sharks, and basking sharks. My coauthors and I have written before about how such a transition from eating shelled stuff to eating plankton may have occurred - think about it, what a weird change!
Who's picking on you? Scale-feeding fishes...
our paper in Royal Society Open on the anatomy of lepidophagy.

A paper by my colleagues and I, including my fabulous undergraduate, Jonathan Huie - has just gone live in the Journal of the Royal Society Open Science. Here's the press release by University of Washington's Michelle Ma:
Scale-eating fish adopt clever parasitic methods to survive
Think of them as extra-large parasites.
A small group of fishes — possibly the world's cleverest carnivorous grazers — feeds on the scales of other fish in the tropics. The different species' approach differs: some ram their blunt noses into the sides of other fish to prey upon sloughed-off scales, while others open their jaws to gargantuan widths to pry scales off with their teeth.
A team led by biologists at the University of Washington's Friday Harbor Laboratories is trying to understand these scale-feeding fish and how this odd diet influences their body evolution and behavior. The researchers published their results Jan. 17 in the journal Royal Society Open Science.
"We were expecting that with this specialized scale-eating niche, you would get specialized morphology. Instead, what you get is a mosaic of strategies for the end goal of scale feeding," said lead author Matthew Kolmann, a postdoctoral researcher at Friday Harbor Laboratories.
"This niche has a hidden complexity to it, and it is yet another story about the incredible diversity of life on Earth."
The researchers compared two species of piranha fish — one that feeds on scales only as a juvenile and another that eats scales its whole life — and two species of characin fish, commonly known as tetras, with similar eating habits as the piranhas. They found that all four of the scale-feeders varied considerably in their body shape and feeding strategy.
The piranha that eats scales its whole life, named Catoprion mento, tends to live alone. When it does hunt, it swims up behind its prey, opens its large, Jay Leno-like jaw 120 degrees and pries large scales off the sides of other fishes. These piranhas can tolerate nearly a dozen large fish scales in their stomachs at one time; that's like a human swallowing a dozen silver dollar pancakes in a single bite.
In contrast, the characin fish most similar to the piranha in life history and eating preferences gets its food in an entirely different way. The blunt-faced fish, called Roeboides affinis, has teeth on its nose and butts its face into other fish, devouring the scales as they fly off from the force of impact.
The research team gathered its data by CT scanning specimens of each fish at different ages at Friday Harbor Laboratories on San Juan Island. Using iodine-contrast staining, they were able to examine the internal anatomy of the four species to better understand what traits are shared by fishes that employ such a rare feeding strategy.
The stark differences in jaw and head shape, combined with how each prefers to hunt, shows a great diversity among the small number of species that have evolved to eat scales, the researchers found.
"This study would have been extremely hard to do without the CT scanning," said co-author Jonathan Huie, a UW undergraduate student in aquatic and fishery sciences. "We were able to look at the image slices from three different points of view, and could more accurately pinpoint and measure certain elements like the jawbones."
About 50 fish species are classified as scale-eaters, and all of them live in the tropics. Previous studies have shown — and the CT scans confirm — the fish are able to digest entire scales. The mucous lining the inside of each scale is thought to be appealing to fishes, but there could be other reasons why they prefer the entire scale, Kolmann said.
Most of the piranha and characin scanning was completed last spring during an undergraduate marine biology course at Friday Harbor Laboratories. Huie, then a student in the class, and the other co-authors scanned fish from collections all over the U.S., then meticulously measured a series of traits that are important for feeding, such as the sharpness and shape of various teeth.
The completed scans join a growing online library of 3-D digital fish replicas, pioneered by Adam Summers, a co-author and professor at Friday Harbor Laboratories, with the intent of creating an open-source repository for researchers and the general public to learn about all of the fish on Earth.
Co-author Kory Evans, a postdoctoral researcher at the University of Minnesota, and Kolmann conceived of this project when they both discovered rare, scale-eating fish in their respective field projects involving different fish species in the Amazon.
Kolmann will continue studying the evolution and feeding patterns of piranhas in his new position at George Washington University, where he will have access to the Smithsonian Institution's fish collections.
This research was funded by Friday Harbor Laboratories scholarships and fellowships.
There be sharks here, cap'n!
our paper in Neotropical Ichthyology on shark biodiversity in Guyana.

Q: Where is Guyana?
A: Guyana is a county on the northern coast of South America, flanked by Venezuela to the west, Suriname to the east, and Brazil to the south. Parts of these four countries are collectively called ‘the Guianas’ - because of their peculiar ‘tepuis’ or tabletop mountains - the remains of very ancient rock formation called the Guiana Shield. Guyana is also one of the few non-island Caribbean nations, and it is the only South American country where English is the official language.
Q: What’s so special about Guyana (and the Guianas)?
A: Biodiversity is incredibly high in the Guianas - and not only is it high, animals and plants from the Guianas are very distinct from their cousins in the Amazon and the Orinoco regions. The long existence of the tepuis have isolated Guiana wildlife from other regions, making many species here ‘endemic,’ i.e. found nowhere else in the world. Sir Arthur Conan Doyle recognized the rare and diverse quality of Guyanese wilds and set his ‘Lost World’ there accordingly. A recent study by my collaborator and mentor, Dr. Hernan Lopez-Fernandez, found that 30% of the fish genera in one Guyanese river are found only in that one river.
Q: Wait, so far you’ve talked about freshwater fish - what about the oceans?
A: Dr. Hernan Lopez-Fernandez and many other scientists, after decades of field expeditions, are finally getting an understanding of what freshwater fish species are found in Guyana, although there are literal lifetimes left of work to do, describing, cataloging, and understanding the biodiversity in the Guianas. But we know very little about marine life in Guyana, with most of what we know coming from industrialized fishing fleet records from Venezuela. What we do know is that the marine ecosystem is presumably diverse, with plenty of large animals like turtles, manatees, dolphins, and sharks occupying coastal waters. Since studies in the 1950s by Dr. Rosemary Lowe-McConnell, there have been exceedingly few studies on Guyana’s marine life.
Q: So, what about sharks in Guyana? How could no one know what species are there?
A: Our paper in Neotropical Ichthyology used DNA barcoding, which analyzes fragments of the genetic code that are unique to each species, to identify what sharks are caught in Guyana. There is a coastal, directed driftnet fishery for sharks in Guyana - long floating monofilament nets (floating gillnets) catch large and small coastal species alike, but when these sharks are caught, their heads are chopped off (and sometimes their fins) to make room for more sharks to be transported to market. Like many places in the world, the government has a tough time tracking how many sharks are caught and what species, particularly because landed fish are usually decapitated. My Guyanese collaborator surveyed these coastal markets and sent us small bits of shark fins to use as genetic samples.

Q: What sharks are caught in Guyana? What is the weirdest/coolest species you found?
A: We identified 14 shark species in Guyanese coastal markets (as well as several species of rays identified during other trips). Most of these species are well-known throughout the Caribbean, like Bull Sharks (Carcharhinus leucas), Blacktip Sharks (C. limbatus), and Hammerhead Sharks (Sphyrna spp.). A sizeable proportion of the catch were smaller species well-known to any coastal anglers in Florida, Alabama, or elsewhere: Sharpnose Sharks (Rhizoprionodon spp.). Our Guyanese collaborators reported many Scalloped and Great Hammerhead sharks, worrisome because these two species are threatened by coastal fisheries, and are listed as Endangered by the International Union for Conservation of Nature (IUCN). We also found putative evidence of the Scoophead Shark, Sphyrna media. I say ‘putative’ because samples of this species aren’t found in genomic databases, but the sample was highly divergent from other hammerhead species (and we have picture of this specimen!).
Q: What got you interested in Guyana? When did you first go there?
A: I did my doctoral work at the University of Toronto in Ontario, Canada. I worked consistently with the Royal Ontario Museum’s ichthyological collections, bringing specimens back to the museum. Dr. Lopez-Fernandez sent me on numerous expeditions with Dr. Donald Taphorn, a ROM research associate, to Guyana surveying fish biodiversity with World Wildlife Fund in 2013. It was during these trips that I met Elford Liverpool, and we went visiting coastal fish markets just to sate our curiosity about what species were being caught. Elford and I realized that there was very limited information on what sharks were caught in Guyana, and we had a challenging time identifying some market specimens confidently without their heads attached. Guyana is a fabulously beautiful place and it is a veritable wonderland for an ichthyologist.
Some stingrays have extra-jointed mouths
our recent paper in the Zoological Journal of the Linnean Society

The Neotropical freshwater stingrays (Potamotrygonidae) are ecologically diverse, invading South America from the Caribbean some 30-40 million years ago. Since this invasion these stingrays have diversified to fill myriad niches, with some species eating only fish, others consuming shrimp and crabs, and the strangest perhaps, are those rays which eat aquatic insects (see below, Kolmann et al., 2016). There ecological diversity mirrors some considerable anatomical diversity and in particular one very, very strange thing about potamotrygonids... they have an extra joint or hinge in their skulls.
These freshwater rays have a skeletal element, the angular cartilage, that runs between the hyomandibulae and the jaws. The hyomandibulae extend the jaws away from the head when sharks and rays feed, protruding the jaws away from the skull. The angular adds an additional point of rotation to the feeding skeleton, like adding another elbow or a knee to your arms or legs. Why? Well we discuss in our paper the potential origins of this structure, how it varies across the freshwater ray family, and explore the ramifications of its structure and function.
Turns out these rays can have more than one angular cartilage, with some species having up to three! Other taxa have none or may have even lost the angulars they did have. But why?
For rays that are eating fishes and for those stingrays crushing prey like big crabs and mollusks, we think they need particularly rigid jaws, i.e. jaws without an extra element like the angular cartilage(s). But for those rays which are actively chewing their prey a lot, these species tend to have particularly long angulars, giving their jaws extra range of motion.
The angular cartilages are a synapomorphy (unifying trait) for the family Potamotrygonidae, and have been lost in Paratrygon and Heliotrygon, the piscivorous freshwater rays. These skeletal cartilages can be added or lost sequentially, but we find no evidence that rays can start with multiples (e.g. three) and then lose all those cartilages at once, rather rays lose a single angular cartilage at a time. After surveying the literature, we pose the hypothesis that the angular cartilages evolved as a fibrocartilage - the cartilage equivalent of sesamoid bones (e.g. the patella). Download here and see for yourself!
Sharks shift their spine into high gear!
our recent News & Views article in Nature magazine

We discuss in this latest New & Views articles the findings of a recent paper by Marianne Porter and colleagues, who studied how shark vertebral morphology aid in making for more efficient swimming. Sharks can alter the frequency by which they pass waves of motion down their body, which in turn changes how the cartilage in the spines of these animals behaves. The gradient mechanical properties from the intervertebral disks to the vertebrae themselves store and release elastic energy - meaning these sharks have a continuously-variable transmission. Sharks can shift from low-energy cruising to high-speed swimming almost instantaneously, a handy trick for catching prey over a reef or in the open ocean.
‘Stingrays chew like goats’
our paper has been accepted in Proceedings of the Royal Society!

07/09/2016 -
So our (Nate Lovejoy, Ken Welch, & Adam Summers) most recent paper, is about to be released in Proceedings of the Royal Society: Part B and it's getting a bit of press. So I'm giving ya'll the lowdown on what we found and why it's so interesting (before you hear elsewhere).
Here's the gist:
Freshwater stingrays are found in South American river basins and feed on a diverse array of prey. Many of these species specialize on a single kind of prey, be they fish, crustaceans, snails, or even aquatic insect larvae. But not all of these prey are created equal – some prey are harder, softer, or tougher than others. Insectivorous freshwater stingrays are the only elasmobranchs (sharks and rays) to feed on insects – which are difficult to eat and digest due to high amounts of chitin in their exoskeletons, a remarkably complex and tough material. Other vertebrates, namely mammals like shrews, bats, and tenrecs also eat insects and they use complex jaw motions – chewing – to shred chitin to allow digestive juices to breakdown prey. Stingrays can protrude their jaws away from their skull as well as protrude these jaws laterally, to the left or right. Using high-speed videography we determined that stingrays do actually chew their food – just like mammals. We also found that these stingrays lift their disk to suck prey underneath the body – thereby capturing food with their pectoral fin ‘limbs.’ This decoupling of behaviors, prey capture and prey processing, is reminiscent of what is seen in several major radiations of bony fishes where the oral jaws suck prey and jaws in the throat (pharyngeal jaws) crush prey.
Q. There are freshwater stingrays?
A: There are many! You can find freshwater stingrays in many tropical rivers – in North Australia, New Guinea, Thailand, and even West Africa, to name a few places. But the largest concentration of freshwater rays are found in South America – from Guyana and Venezuela in the north, through Brazil and Peru, and even down into Argentina and Uruguay. These rays (Potamotrygon) were originally marine, but invaded South America millions of years ago, back when the Andes hadn’t risen and the middle of the continent was filled by a shallow, slightly salty lagoon, called the Pebas Mega-Wetland (the best name for a big salty swamp that I can think of). They must have been successful, because now there’s over 30 species of freshwater rays in South America – all inhabiting freshwater.
Q. Why is chewing in rays important?
A: Some of these freshwater rays (potamotryonids: literally – ‘river rays’ in Greek) are unique in that they eat aquatic insect larvae – the juvenile phase of insects like dragonflies, beetles, and caddisflies, which live along the bottoms of rivers and ponds. Insect larvae are made of chitin – a tough substance like plastic that has to be sheared or shredded apart – that’s why stingrays chew, to tear chitin into bits. Aquatic insects are quite plentiful, but they are tough to eat – so stingrays that chew have found a means of eating a plentiful food option that they don’t have to compete with other animals to eat!
Q. I don’t understand why you think the comparison to goats is interesting. Why goats?
A: For a very long time, scientists thought that only mammals, like goats, could chew. Some of the earliest mammals fed on insects, and these sorts of critters were the land animals that ‘inherited the earth’ after the dinosaurs went extinct. Chewing is widely believed to have been a real important adaptation that helped mammals take advantage of new diets when they diversified some 60-70 million years ago. What we’re finding more recently is that other organisms that we wouldn’t have expected to be able to chew, are arguably doing the same thing as mammals – and they’re chewing because this behavior seems to be the best solution for eating tough substances like insects, grasses, and even bone. Now we know there are carp that chew. Lizards that chew, even some dinosaurs are hypothesized to have been chewers. But chewing has never been observed in sharks and stingrays, and we’re the first to find it.
Q. Why don’t other stingrays eat insects?
A: Well, we think that to chew on insects you need really flexible jaws. Most stingrays have these, although some of the rays I’ve studied previously, ones that eat mollusks have very rigid jaws. So those clam-eating stingrays probably can’t chew, just crush. Freshwater rays have particularly kinetic, or mobile jaws – the jaws of all rays are extended away from the skull during feeding by two cartilages, the hyomandibulae. But what makes freshwater rays so unique is that they have an extra joint that runs between the hyomandibulae and the jaws – making their jaws extra flexible. But maybe the answer to why more stingrays don’t eat insects in that because there just aren’t that many aquatic insects in the oceans – so only freshwater rays have had the opportunity!
Q. Why would anything eat an insect anyway?
A: Insect larvae are packed with nutritious fats that they’re saving prior to metamorphosing – when these insects change from generally flightless juveniles to volant (flying) adults. All these fat stores are very nutritious and freshwater stingrays sure seem to love them! When I was conducting my experiments, the rays would ignore other foods if I dropped insects into the tanks – even though they took more of an effort to eat!

02/09/2016 - I'm excited to say that I have passed my PhD final examination and I will be starting my post-doc at Friday Harbor Labs in just a few weeks!
Stay tuned for more updates... there are several things cooking in the meantime!

10/07/2016 - Still recovering from the shock, elation, and honor at being awarded the American Elasmobranch Society (AES) Gruber Award at this year's Joint Meeting of Ichthyologists and Herpetologists in New Orleans.
I've had some really tremendous influences and mentors along the way, and I in particular thank my coauthors, Nate Lovejoy, Adam Summers, & Ken Welch for their support and advice along the way. Couldn't have done it without these gentlemen scientists.
Sonja Fordham (Shark Advocates) - is credited for snapping this photo of me with past winner Chris Mull and sawfish whisperer, Dana Bethea! Great seeing you two again!

04/07/2016 - The International Congress of Vertebrate Morphology was a whirlwind of activity and I barely could keep up with all the sorts of cutting edge science taking place!
Particularly fascinating with Mason Dean et' al's symposium on biomaterials research - such absolutely awesome stuff! Highlights were biomimetic composites that simulate nacre and glass sponge spicules 3D printed at Harvard by my collaborator James Weaver!
In case you didn't get a chance to see it, check out my poster to the right on the evolution of insectivory in freshwater rays - a curious story of a 'lower' vertebrate which effectively chews tough prey like aquatic insect larvae. Insect larvae are widespread in tropical and temperate freshwater systems - but are a dietary niche that has remained out of reach for most elasmobranchs (sharks and rays) until now.

16/06/2016 - The folks at Experiment.com contacted me regarding creating a project for crowd-sourcing through their Shark Research Challenge, during Shark Week.
Please visit my research page there and consider donating (we're about 20% there after a week!) or share widely among your friends and family.
I'm trying to determine just how widely among the freshwater rays complex feeding behaviors, like chewing, are distributed and how these behaviors correlate with their dietary ecology and morphology.
Maybe most importantly for the casual reader, I've written several 'LabNotes' for this project which introduces you to the freshwater ray 'phenomena' - why they're so interesting, where they come from, and how my research is answering these sorts of questions (and generating more questions, too).
Alternatively, check out my blog post on Southern Fried Science, too! If anything - it's a fun read!

01/06/2016 - Our JEB paper, 'Morphology does not predict performance' was chosen by the UToronto Scarborough Biology Dept as the best student paper of 2016! Nice way to enter into the summer stretch towards dissertation defense!
Thanks to my coauthors, Nate Lovejoy, Mason Dean, Adam Summers, and Stephanie Crofts!
My undergraduate research assistant, Swara Shah - also one best undergraduate student research project for her research with me on bullnose ray feeding biomechanics!
Congrats Swara!

15/04/2016 - I was interviewed regarding the talk I gave at the 2016 SICB meeting in Portland OR on how stingrays feed on tough and stiff biomaterials. So how do they do it? Well, not to give away too much, rays feeding on tough prey have incredibly flexible, many-jointed (kinetic) jaws that help them "chew" prey like insect larvae and crustaceans. Rays that specialize on stiff prey like mollusks, on the other hand, use a drastically different strategy - fusing their jaws and teeth into a single, solid crushing platform. The diversity of feeding modes in stingrays is impressive given their low species richness (# of species). I thank the writer, Kaitlyn Lowder, for doing such a great job!

07/04/2016 - My BIOC99 (Seminar in Biomechanics and Comparative Phylogenetic Methods) presented their work as posters to our lab group today - and did a fantastic job. Swara presented work she did with another student of mine, Henil Patel, on scaling of feeding performance in bullnose rays (Myliobatis freminvillei). The highlight of this study is that these animals have comparable performance to other durophagous rays, but it is driven by efficient force transfer rather than increases in gross muscle morphology, as we documented in related cownose rays. Moreover, a sesamoid-tendon complex in these animals keeps pace with muscle growth and jaw leverage in order to offset internal tendon stresses during biting.

Amy & Ereny discussed their findings on functional redundancy and morphological diversity in the jaws of dasyatoid stingrays (below). They found that not only is morphology redundant when its comes to function (i.e. there are diverse shapes, but only several functional outcomes), but performance in these rays increases at a greater rate than one expects across the phylogeny of these animals. Incidentally, freshwater stingrays occupy a comparably huge area of morphospace compared to other rays.

07/01/2016 - Made an early departure from the Society for Integrative and Comparative Biology meeting in Portland, OR. It was a great time, and I got to witness some great science. Good times meeting old friends, and new - but most importantly I got to see my students get their first taste of a professional conference. Swara and Henil did a fantastic job presenting their research! You can see a photo of their poster to the left and the slide from my presentation on prey materials and their influence on stingray feeding morphology (below).

2/12/2015 -
Our new paper featured in Journal of Experimental Biology went live - here's an FAQ about what we found and why it's interesting!
Q. What is the most important point of the paper?
A: The jaws of hard prey crushing stingrays have really different shapes. Despite this no shape is better than any other at smashing hard prey. There are many types of hammers and they all perform equally well. This is unusual because we expect to see performance differences when there are large differences in shape. Consider for comparison the beaks of Galapagos finches - different shapes are better for dealing with different types of seeds.
Q. How hard can a stingray bite?
A: Very, very hard...a cownose ray just 2 feet across can generate more than 110 pounds of force. A big eagle ray could generate over 1000 pounds.
Q. Why did you use models of prey items?
A: Nature is messy. Using models reduces the “noise” from natural variation. We used 3D printed shells that were identical in shape and structure to live shells. These faux shells broke in the same manner as live shells. This suggests that shape was not very important in predicting when any shell would be destroyed… but what the shells are made of… well that was pretty important!
Q. If shape is not important why does it vary?
A: Shape CAN be important, if it artificially inflates the size of the prey. Prey can act larger by having large spines or knobs or ridges. Predators must then be large enough to swallow the whole prey, including the spines. Spines discourage smaller predators, and large predators if the spines are venomous or sharp.
Q. Are there any of these stingrays near where I live?
A: Most of these rays are tropical, with cownose and bullnose rays occurring as far North as Delaware Bay in the United States. In the Caribbean, Florida, and the Bahamas you can find spotted eagle rays (Aetobatus). But other types of eagle rays (Aetomylaeus) are found only in the Indo-Pacific, alongside the other three stingrays.
Q. Is crushing hard prey common?
A: There are many critters that crush hard prey, hyenas, for example. All are very specialized, with particular teeth, jaws, and big muscles. That sort of suggests a common evolutionary theme that molds these creatures along similar processes. Interestingly, there are several, unrelated hard-prey crushing stingrays, despite their having a cartilaginous skeleton.
05/11/2015 -
Jillian Morris featured some of my batoid-related research interests over at Sharks4Kids! In case you haven't heard, Sharks4Kids teaches young people about ocean life, especially elasmobranchs, in order to inspire the next generation of shark advocates through education, outreach and adventure.
11/11/2015 -
Bit of departmental recognition for graduate students here, one of which was me and my buddy Derrick!

02/11/2015 -
There's been a lot of controversy about the collection of the Moustached Kingfisher, an infrequently-collected species from Guadalcanal in the Solomon Islands (https://en.wikipedia.org/wiki/Guadalcanal).
I'll leave the story to the news article, but when the collecting scientist, Dr. Filardi of the American Museum of Natural History wrote an op-ed to defend his collection of the animal, he was lambasted for being a "murderer" amongst other things. I tried to explain why collections are still absolutely necessary in today's world and some of my comments are featured in this great summary article by Audubon:
...what I've been doing at Harvard.
HARVARD, PEOPLE... HARVARD

26/07/2015 -
The time is nigh... I begin my lengthy roadtrip from UToronto to Academy of Natural Sciences (ANSP) to grab specimens and then onward to Cambridge MA to start my research position at the Wyss
Institute at Harvard University. I'm incredibly humbled to have been given this opportunity.
I will be ct scanning dozens of species of stingrays. There are two over-arching goals here, (1) to generate a dataset for my last doctoral dissertation chapter on morphological evolution of the feeding apparatus in marine and freshwater stingrays and (2) gather more data for Mason Dean and James Weaver's project on the mechanics of tessellated cartilage. I'm stoked.
That big brown floppy thing in the photo is a torpedo ray, just fyi!
22/07/2015 -
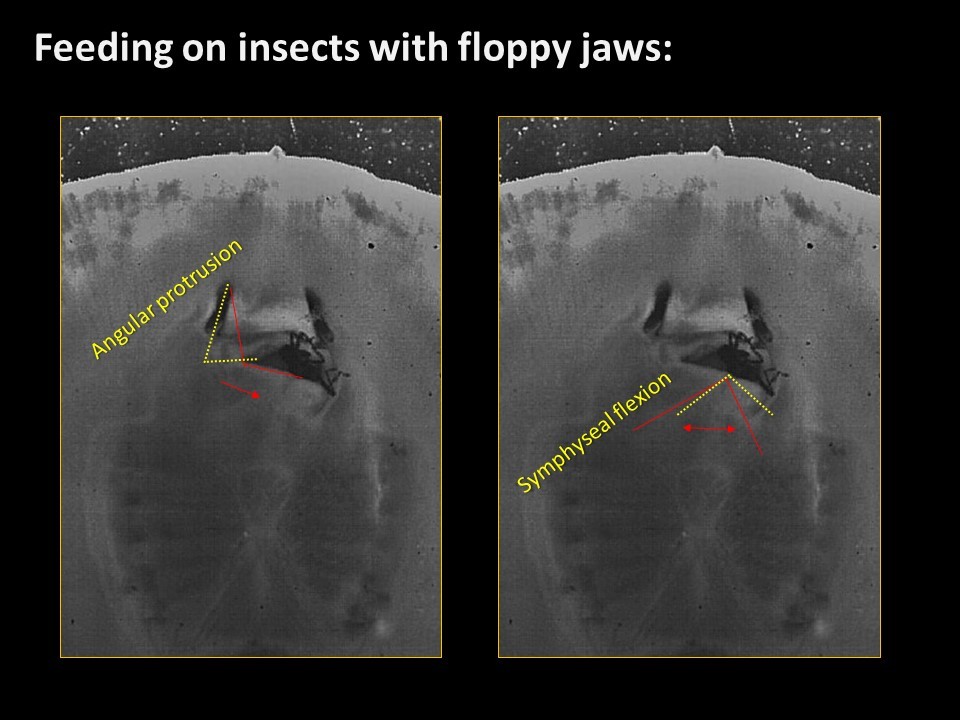
If you missed my talk @ AES/JMIH 2015 - you can see a sampling of the slides below. Turns out asymmetrical jaw protrusion is a pretty common behavioral condition of prey capture in Potamotrygon. Why? Well we're not sure yet - but maybe it has something to do with shearing insect prey.
Kolmann, M.A. & Lovejoy, N.R. Feeding kinematics of the ocellate freshwater stingray, Potamotrygon motoro. American Elasmobranch Society. July 2015. Reno, NV.
and be sure to contact Lisa Whitenack for information regarding our study on the functional morphospace of shark teeth since the Permian(!):
Whitenack, L.B. & Kolmann, M.A. Integrative Chondrichthyan Paleobiology: The Present is the Key to the Past. American Elasmobranch Society. July 2015. Reno, NV.
27/05/2015 -
Great news - our paper on the scaling of bite force generation in cownose rays for the Journal of Anatomy has been accepted! Check out my ResearchGate profile soon for a copy!
How "hard" can a stingray bite? Think you know?
(and by "hard" I mean "forcefully")

5/04/2015 -
Happy Cinco de Mayo! See below for slides from my presentation at the Interdisciplinary Approaches to Fish Skeletal Biology in Tavira, Portugal <http://iafsb.org/>!
I had a great time and met some fantastic people. Not to mention the fish markets were great for specimens (Engraulis and Torpedo, nice!) My talk focused on our meager understanding of how durophagous stingrays use mineralized teeth, stress-dissipating struts, and massive muscles to process prey like shellfish. I found that jaw shape has no effect on crushing performance and muscle morphology is the primary contributor to positive gains in feeding performance in these wild critters!
Kolmann, M.A., Summers, A.P., Lovejoy, N.R. Development and decoupling of form and function in the tooth plate modules of durophagous stingrays. Interdisciplinary Approaches in Fish Skeletal
Biology. March 2015. Tavira, Portugal.
Marowijne & Corantijne Rivers, Suriname
17/04/2015 -
Been a little while on updates - unless you've been following me on Twitter @KolmannMA.
The ROM expedition to the Marowijne and Corantijne Rivers was a success, as was my own expedition to the Lower Demerara, to survey estuarine and freshwater fishes.
Not only did my collaborators and I collect ~115 spp in 44 families, including stingrays for my thesis - but in Suriname we re-discovered a species from it's type locality that hasn't been seen in over 60 years, a banded knifefish (Gymnotus anguillaris).
So what does this mean exactly? Well, when a new species is discovered and described, several specimens are put aside as "types" - holotypes and paratrypes - and are distributed between several museums. These type specimens embody what is that species - a voucher that is available for reexamination, sort of like a entry in a glossary (but an exceedingly dead fish). With the advent of modern molecular methods in systematics, fresh tissue is needed to provide a biochemical "type" specimen (along with the preserved specimen being a morphological type), what we call a tissue voucher.
Since this banded knifefish was discovered (like many species) before the genesis of molecular phylogenetics (methods of determining evolutionary relationships from patterns of DNA) researchers lacked any record of its genetic distinctiveness. Now we have a record of this fish, from a molecular standpoint - and this has allowed us to determine exactly which species of knifefish this particular fish is related to - and how they have dispersed themselves across the Guianas.
Lower Demerara River, Guyana

This last March I led a sort of hybrid expedition/fisheries dependent survey of the Lower Demerara and its estuary, funded by Rufford Foundation. We went fishing with locals, intent on determining which fishes were being caught for local consumption and export - especially what species of sharks and rays were being harvested. So we get to analyze not just the local biodiversity, but also the "ecology" of the fishermen... with some interesting results. The highlight of the trip for me is catching the long-snout stingray Dasyatis geijskesi... a little known component of the ichthyofauna (fish diversity) in Guiana Shield estuaries. I'm hoping to start a project with the University of Guyana on understanding the reproductive biology of this stingray, which seems (anecdotally) to be strange, rather slow for a dasyatid.
23/02/2015 -
Had a great time at Cano Palma Biological Station with a group of stellar students: http://www.coterc.org/cantildeo-palma-overview.html
Cano Palma is a field station organized by the Canadian Organization For Tropical Education and Rainforest Conservation (COTERC). The station generates baseline survey data for shorebird migrations, turtle nesting, snake demography, caiman populations, and mammal diversity in addition to being a base for visiting scientists pursuing their own research year-round. The station has hosted the Lovejoy Lab's Tropical Biodiversity course for two years now and the area features an interesting array of fishes - from characids to poeciliids!
All fishes are being identified and taken to the Royal Ontario Museum where they will be some of the first records of the fish fauna inhabiting northeastern coastal Costa Rica - in fact, we may have a new species or two, which is pretty exciting!
We caught some great fishes and overall, had ourselves a time! But I miss my station people...
**Update** and we got featured in the University news!:
http://news.utoronto.ca/monkeys-alarm-clocks-biodiversity-expedition-tropical-rainforest
some of the photos below courtesy of Patrick Traynor
10/01/2015 -
Got back from SICB 2015 (http://www.sicb.org/meetings/2015/) and caught up with my FHL friends and collaborators! It was a hell of a meeting - accelerometers, suction-feeding, and ballistic tongue projection - oh my... For those of you who may have missed it - here's a poster of the work I did at FHL and presented at SICB in West Palm Beach.

10/12/2014 -
Just a few photos of me filming Potamotrygon motoro feeding using high-speed videography - we're trying to figure out how these animals use de-coupled (loosely-connected skeletal structures) to feed on complex prey!

01/11/2014 -
Donald Taphorn and I just received a grant through the Rufford Foundation to study freshwater and estuarine fish biodiversity in the Demerara River, Guyana!
We will be working with local fishermen and recruiting a local University of Guyana student to catalog commonly-caught fish species as well as document fish in the Demerara, probably the most
extensive scientific survey in this river for a hundred years! Hopefully we'll catch more Potamotrygon too!

08/10/2014 -
Back to Friday Harbor to continue my research on shell mechanical properties and how durophagous (animals that eat shelled prey) stingrays crush mollusks!
Here's a Nucella snail about to be crushed using replica Aetomylaeus (Indo-Pacific eagle ray) jaws made from ct scanned cross-sections and milled from solid aluminum!
More photos below! Skates (Rajidae), snailfish (Liparidae), sunsets, lighthouses, varnish clams (Nuttalia), 3D printers and mechanical loading frames (MTS Corp).
30/07/2014 -
Got finished with Adam Summer's Functional Morphology of Fishes course (I highly recommend):
http://depts.washington.edu/fhl/studentSummer2014.html
http://faculty.washington.edu/fishguy/People/Adam-Summers-Teaching.html
And I already miss Friday Harbor! Not only did I learn how to identify a whole series of fish families new to me, being a temperate/tropical United States coastline person, like cottids and embiotocids - but I learned a ton of new stuff! Reconstructing ct scans, rendering images in 3D CAD software, 3D programmable milling (stingray jaws out of aluminum!), and 3D printing - one hell of a class!
Pictures below!
05/2014 -
Eigenmann in the early 1900s surveyed the fishes of Guyana, but only in two places - the Essequibo and the Potaro rivers. His lists of fish and their species descriptions are some of the
most accurate for this entire region of the Guiana Shield, a region of uplifted Precambrian rock that inspired Sir Arthur Conan Doyle to write the Lost World. We didn't find any dinosaurs,
but every time we come to Guyana we find new species!
This time in Guyana, myself, Donald Taphorn, Hernan Lopez-Fernandez, Jon Armbruster and others surveyed the Potaro, Kuribrong, and Amaila rivers for a potentially threatened fish new to science,
Characidium amaila, a strange crenuchid (Crenuchidae) fish from the lower Amaila River. We not only found the fish, but we also are the first biologists since 2004 (and before
that, 1904) to find and sample the sister taxa to the armored catfishes (Loricariidae) - Lithogenes villosus! This strange little fish is unarmored, unlike it's relatives and uses
its suckermouth to attach to rocks in high-flow rapids (rheophilic). Always a pleasure to work with World Wildlife Fund in this beautiful country.

01/03/2014 -
Greetings from the Ucayali, Peru!
We're outside of the Pacaya-Samiria Biological Reserve sampling for knifefishes (Gymnotiformes)... these fishes produce an electric signal (EOD) through a modified muscle-structure called an electric organ (EO). Knifefishes use this signal (a wave or pulse) to navigate their environment, communicate with other knifefishes, and in the case of the electric eel (Electrophorus) actually shock predators and prey! We're trying to examine the genomic basis of electric signal differentiation between the sexes by recording the EOD from different species of knifefishes and analyzing the patterns of gene expression in EO tissue.

10/30/2013 -
World Wildlife Fund Guianas (WWF) contacted the Royal Ontario Museum (ROM) to help them survey fish biodiversity (as part of a larger, multi-taxon biodiversity survey or BAT) in the Lower
Rupununi river basin in Guyana. Hernan Lopez-Fernandez (Curator of Fishes at the ROM) sent me to Guyana with his mentor, Dr. Donald Taphorn to work with WWF. What an experience!
Guyana is such a beautiful place - full of fishes and other interesting critters. A growing concern there is the expansion of artisanal gold mining along the rivers - which lead to
erosion and leaching of mercury into the water system - harmful for local wildlife and native peoples alike. With pressure from the rest of the world for cheaper gold, miners are moving
into the pristine interior of Guyana's waterways to strip mine the area. We found a remarkable array of fishes and other wildlife, now cataloged at the ROM, with some new species awaiting
description. More photos can be found under the Fieldwork tab. I wonder if I can add anymore acronyms to this paragraph...


16/04/2013 -
New things to come now that I have secured independent funding for investigating the functional and evolutionary ramifications of cranial muscle evolution and jaw suspension in chondrichthyans. Expect to see how potamotrygonid stingrays (Family Potamotrygonidae) and allies can help us understand how molecular evolution, physiological redundancy and functional modularity has taken us vertebrates from our jawless ancestors to our shiny, toothed gnathostome heritage!
Also, look forward to new information on how stingrays have evolved to fill niches, new ecological roles and may be converging on similar morphological strategies... another example of freshwater fish and adaptive radiation?

12/21/2012 - UPDATE:
Check out the new updates in the Research section as I finally get the opportunity to start publishing my MSc work and start geeking out about my dissertation potential!
Changes to the About Me and Lab sections as well!
Stay tuned, lots of changes coming up - especially my new crowd-sourcing section!
Teaching in Costa Rica to commence in February! Back to the Tropics!

07/30/2012 - UPDATE:
Well, I'm now officially a Master of Science! Starting my PhD at UToronto in the Lovejoy lab on neotropical fish diversity, biogeography and speciation!
On to Potamotrygonidae!

Summer - UPDATE:
Getting ready to defend the MSc, couple last minute revelations about Rhinoptera, but maintaining sanity with field work in the Bahamas (Andros), the Florida Keys (pictured), and the Lost Coast of FLA (Steinhatchee fish fry, here we come!).
Come see my talk at the Joint Meeting of Ichthyologists & Herpetologists (AES/ASIH) in Vancouver this year!
 Kolmann Lab
Form.Function.Evolution
Kolmann Lab
Form.Function.Evolution